Unveiling the Power of LC-MS Applications in Drug Development

4 out of 5
| Language | : | English |
| File size | : | 3885 KB |
| Text-to-Speech | : | Enabled |
| Print length | : | 256 pages |
| Lending | : | Enabled |
| Screen Reader | : | Supported |
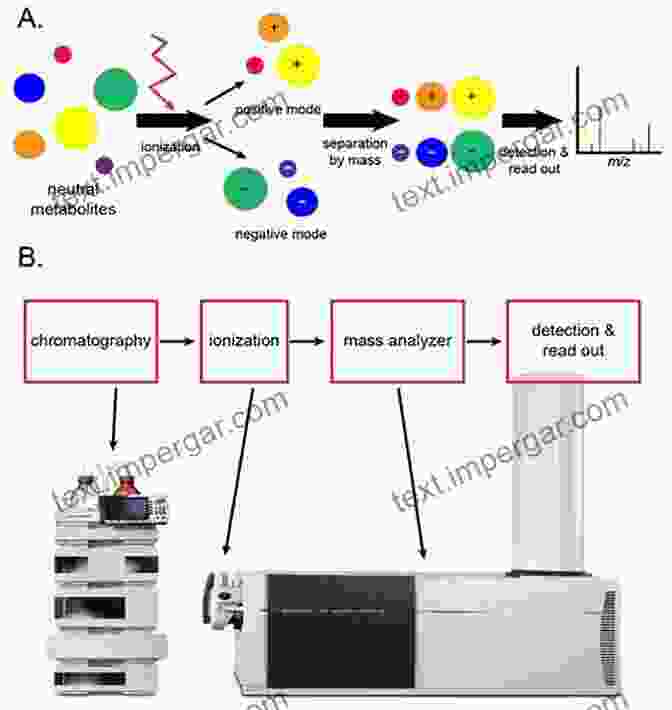
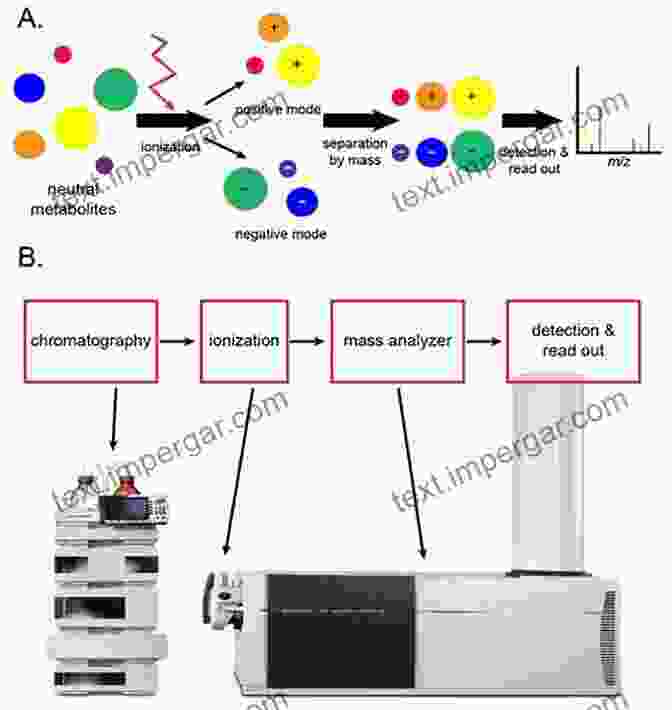
Mass spectrometry (MS) has revolutionized various scientific disciplines, including drug development. Liquid chromatography-mass spectrometry (LC-MS) is a powerful analytical technique that combines the separation capabilities of liquid chromatography with the mass-analyzing capabilities of mass spectrometry. This unique combination enables scientists to identify, characterize, and quantify compounds in complex mixtures, making LC-MS an indispensable tool in the drug development process.
LC-MS in Drug Discovery
In drug discovery, LC-MS plays a crucial role in identifying and characterizing potential drug candidates. By analyzing samples from biological assays, LC-MS can detect and measure the presence of specific compounds, providing valuable information about their activity and selectivity. Additionally, LC-MS can be used to optimize drug properties, such as solubility, stability, and absorption, by evaluating the impact of structural modifications on these parameters.
LC-MS in Drug Optimization
Once a drug candidate has been identified, LC-MS is employed to optimize its properties and ensure its safety and efficacy. By analyzing drug metabolites, LC-MS can provide insights into the drug's metabolism and clearance pathways, enabling scientists to identify potential drug-drug interactions and optimize dosing regimens. Additionally, LC-MS can be used to evaluate the stability of drug formulations and monitor drug levels in patients, ensuring the drug's safety and effectiveness.
LC-MS in Drug Safety Evaluation
LC-MS plays a critical role in evaluating the safety of drug candidates. By analyzing samples from toxicology studies, LC-MS can identify and quantify potential toxic metabolites and assess their potential to cause adverse effects. Additionally, LC-MS can be used to monitor drug levels in patients during clinical trials, ensuring the drug's safety and efficacy.
Best Practices for LC-MS in Drug Development
To ensure the accuracy and reliability of LC-MS data in drug development, it is essential to follow best practices throughout the analytical process. These best practices include:
- Proper sample preparation and extraction techniques
- Optimization of LC separation conditions
- Careful selection of MS ionization and detection methods
- Validation of analytical methods
- Quality control measures to ensure data integrity
LC-MS has become an indispensable tool in drug development, providing scientists with a powerful means to identify, characterize, and quantify compounds in complex biological samples. Its applications span the entire drug development process, from drug discovery to optimization and safety evaluation. By harnessing the full potential of LC-MS, pharmaceutical scientists can accelerate drug development and deliver safe and effective therapies to patients.
References
- LC-MS in Drug Development: A Practical Guide, John Wiley & Sons, 2018.
- Mass Spectrometry in Drug Discovery, John Wiley & Sons, 2015.
- Best Practices for LC-MS in Drug Development, The American Association of Pharmaceutical Scientists, 2019.
4 out of 5
| Language | : | English |
| File size | : | 3885 KB |
| Text-to-Speech | : | Enabled |
| Print length | : | 256 pages |
| Lending | : | Enabled |
| Screen Reader | : | Supported |
Do you want to contribute by writing guest posts on this blog?
Please contact us and send us a resume of previous articles that you have written.
 Book
Book Novel
Novel Page
Page Chapter
Chapter Text
Text Story
Story Genre
Genre Reader
Reader Library
Library Paperback
Paperback E-book
E-book Magazine
Magazine Newspaper
Newspaper Paragraph
Paragraph Sentence
Sentence Bookmark
Bookmark Shelf
Shelf Glossary
Glossary Bibliography
Bibliography Foreword
Foreword Preface
Preface Synopsis
Synopsis Annotation
Annotation Footnote
Footnote Manuscript
Manuscript Scroll
Scroll Codex
Codex Tome
Tome Bestseller
Bestseller Classics
Classics Library card
Library card Narrative
Narrative Biography
Biography Autobiography
Autobiography Memoir
Memoir Reference
Reference Encyclopedia
Encyclopedia Missy Johnson
Missy Johnson Max Wallace
Max Wallace Michael P Lynch
Michael P Lynch Timothy Palzkill
Timothy Palzkill Michael L Eads
Michael L Eads Steven Varnell
Steven Varnell Marcus Garvey
Marcus Garvey Michael Summers
Michael Summers Michael Joseloff
Michael Joseloff Nayan Shah
Nayan Shah Richard A Lovett
Richard A Lovett Myron R Ferguson
Myron R Ferguson Ralph D Sawyer
Ralph D Sawyer Michael Tonsmeire
Michael Tonsmeire Mike Berry
Mike Berry Michael Barclift
Michael Barclift Michelle Higgs
Michelle Higgs Melody Carlson
Melody Carlson Ted Owens
Ted Owens Muskan Jha
Muskan Jha
Light bulbAdvertise smarter! Our strategic ad space ensures maximum exposure. Reserve your spot today!

 Christopher WoodsUnlock the Secrets of Chemical Engineering with Butterworth Heinemann's...
Christopher WoodsUnlock the Secrets of Chemical Engineering with Butterworth Heinemann's... Eddie PowellFollow ·8.5k
Eddie PowellFollow ·8.5k Efrain PowellFollow ·12.2k
Efrain PowellFollow ·12.2k Dwight BlairFollow ·15.8k
Dwight BlairFollow ·15.8k Gabriel Garcia MarquezFollow ·8.5k
Gabriel Garcia MarquezFollow ·8.5k Ira CoxFollow ·2.7k
Ira CoxFollow ·2.7k Edison MitchellFollow ·10.6k
Edison MitchellFollow ·10.6k Frank ButlerFollow ·9.3k
Frank ButlerFollow ·9.3k Bret MitchellFollow ·4.6k
Bret MitchellFollow ·4.6k

 James Gray
James GrayCharles The Bold Illustrated: An Epic Journey Through...
Step into the captivating world of Charles the...

 Harold Blair
Harold BlairUnveiling the Ultimate Guidebook for Commerce...
Embark on a comprehensive journey through...

 Percy Bysshe Shelley
Percy Bysshe ShelleyDitch Dare Do 3D: Personal Branding for Executives
In today's...

 Eddie Bell
Eddie BellProfessional Nursing Practice In The United States: A...
In the dynamic...

 Brenton Cox
Brenton CoxThe Concept of Reduction: A Philosophical Odyssey
The concept of...
4 out of 5
| Language | : | English |
| File size | : | 3885 KB |
| Text-to-Speech | : | Enabled |
| Print length | : | 256 pages |
| Lending | : | Enabled |
| Screen Reader | : | Supported |












